The type of resistance and the speed of resistance development will be affected by population size, genetics, the method of reproduction, generation time of the pest and the level of exposure to the chemical (rate and number of applications).
Population size
It is a numbers game. The greater the population size, the greater the number of organisms that will survive a chemical application and carry a resistance mechanism into the next generation.
Genetics
Weeds, insects and diseases have part of their life cycle as diploids (two copies of genes), and haploids (a single copy of genes).
For weeds and insects, the haploid part of the cycle is contained within in diploid form. In weeds, this is the pollen and ova, and in insects, the sperm and egg. As these diploids have two copies of genes when exposed to chemicals, any resistance could be masked (or hidden) if the resistance gene is recessive or only partially dominant. If reproduction is sexual and cross-pollination occurs, further masking of a recessive or partially dominant resistance gene may occur.
For fungi, the haploid and diploid parts of the life cycle are more independent but with the majority of their life cycle as haploids. As fungi are exposed to chemicals while they are in their haploid phase when resistance develops it can show up quickly as there is only a single copy of the gene(s).
Reproduction
The rate of resistance evolution varies for sexual versus asexual (in insects and fungal pests), or cross versus self-pollination (in weeds).
For asexual reproduction, all the genetic information is from a single parental source and is replicated during reproduction with little chance of any diversity other than through mutation. If that source has resistance, all the offspring will have resistance. Both fungi and many insects reproduce asexually and this can result in a fast increase in resistance build-up after it first occurs.
If an organism reproduces sexually, the offspring have a combination of alleles (the genes for each trait) from each parent. When offspring inherit one resistant allele from one parent and one susceptible allele from the other parent, their resistance status depends on which allele is dominant.
More rarely, both alleles have an impact resulting in intermediate resistance (co-dominance). If the susceptible allele is dominant the frequency of resistance alleles may build-up without being noticed. Introducing non-resistant genes has been successfully used to manage insecticide resistance in cotton by having refuges.
Organisms that reproduce sexually are more likely to develop multi-gene resistance, such as metabolic resistance, while asexual reproduction tends to select for major-gene target-site resistance.
In plants, fertilisation can be via cross or by self-fertilisation. Self-fertilisation is similar to asexual reproduction with the genes all coming from a single parent.
However, in self-fertilisation, some mixing of the genes can occur during fertilisation - causing some different types of offspring.
Organisms that reproduce sexually are more likely to develop multi-gene resistance, such as metabolic resistance.
As with asexual reproduction, target-site resistance has a higher likelihood of developing in self-fertilised species due the limited mixing of the genes.
Generation time
A generation is the time involved in passing genes from parents to offspring. When successive generations of a pest are exposed to a chemical, the proportion of a population with the resistance increases. This is because growers are continually selecting for the resistance genes by eliminating the susceptible proportion of a population, leaving only resistant individuals behind.
When successive generations of a pest are exposed to a chemical, the proportion of a population with the resistance increases.
Fungal pathogens can have as many as 10 generations during a crop and insects as many as five generations. For weeds, there is generally only one generation exposed per year, although there may be several cohorts (germinations) from the seedbank that are exposed. This can delay the observation of resistance in the field with a slow build-up of a resistant seedbank. The downside is that this resistant seedbank will also take time to decrease when the selection pressure is removed.
Rate
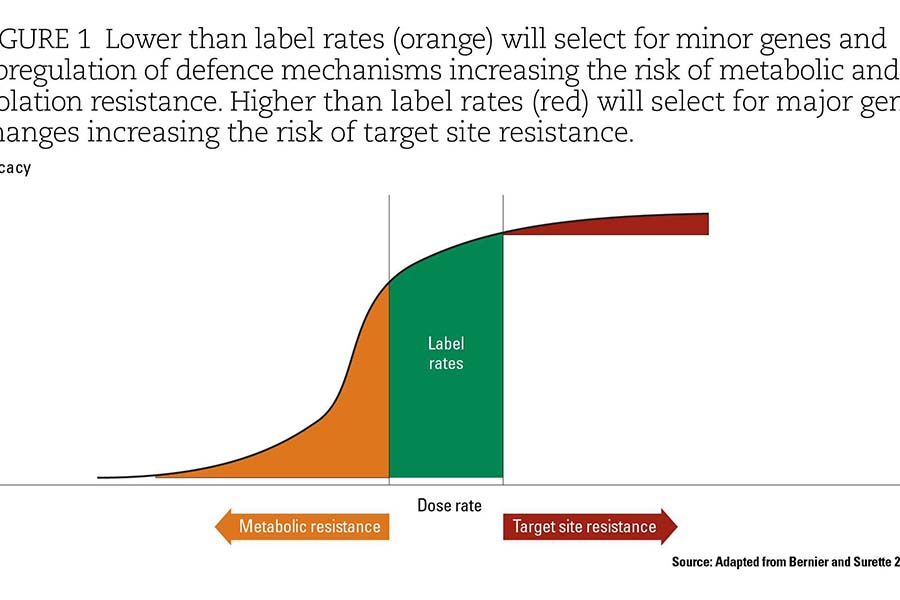
As a broad principle, the use of high rates of an agricultural chemical provides a high selection pressure for major changes to overcome the chemical, such as target site resistance (as shown in Figure 1). Using low rates and sub-lethal rates of an agricultural chemical will cause the upregulation of systems in organisms that can deactivate, isolate or expel the agricultural chemical (that is, metabolic resistance and isolation from target site).
More information: Dr Ken Young, GRDC, 02 6166 4500, ken.young@grdc.com.au
The author acknowledges the assistance of Dr Fran Lpez-Ruiz, Professor Ary Hoffmann and Professor Hugh Beckie.

























































